|
4/1/2024 0 Comments What is sp3d hybridizationIn the C 2H 4 molecule, carbon atoms are sp 2- hybridized and one 2p- orbital remains out to hybridization which forms a p-bond. Formation of C 2 H 4 and C 2 H 2 molecules Therefore, the bond angle in the NH 3 molecules is 107.80. The angle between H-N-H has to be 109.50 but as there is one occupied sp 3- hybrid orbital, the angle decreases to 107.80. Now to form an NH 3 molecule, three 1s- orbitals of three hydrogen atoms overlap with three sp 3 orbitals. 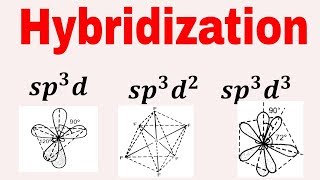
In NH 2 molecule, the nitrogen atom is sp 3- hybridized and one hybrid orbital possesses two electrons. Formation of Ammonia (NH 3 ) and Water (H 2 O) 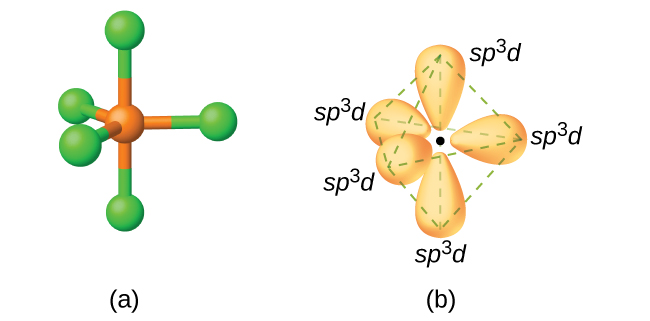
EthaneĦ C-H sigma σ bonds can be formed by the interaction of C-sp 3 with an H-1s orbital and 1 C-C sigma bond can be made by the interaction of C-sp 3 with another C-sp 3 orbital. 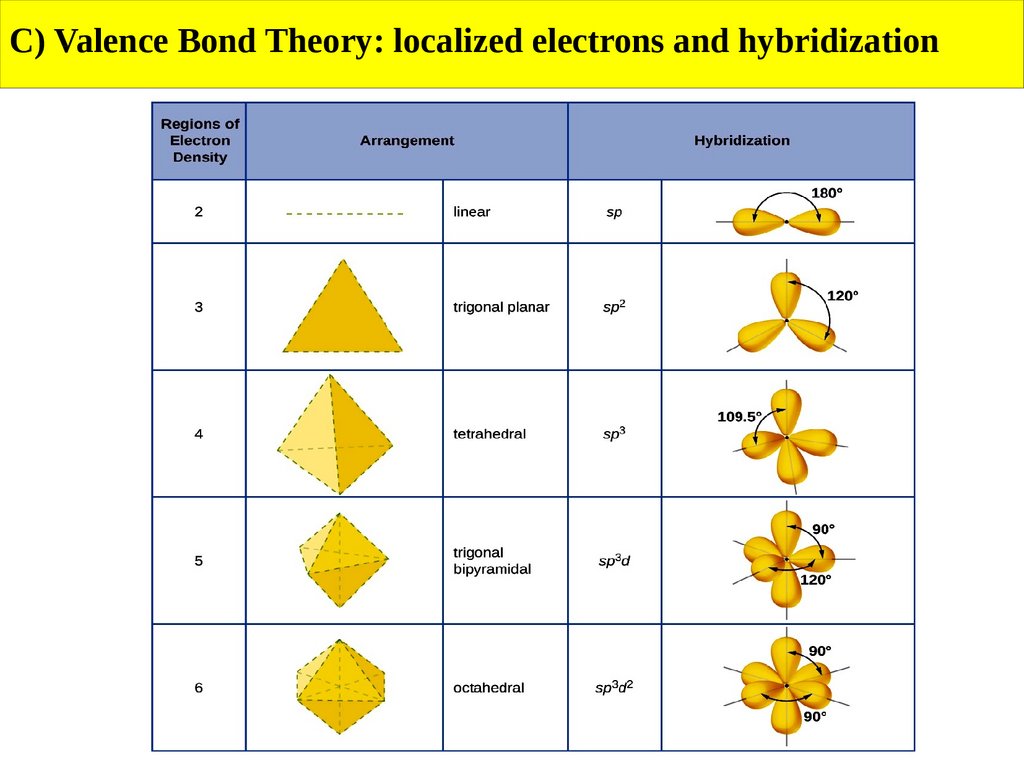
Methaneīy the interactions of C-sp 3 with an H-1s, 4 equivalent C-H σ bonds can be formed.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
